INDIANAPOLIS, March 3, 2023 /PRNewswire/ — Eli Lilly and Company (NYSE: LLY) today announced that the U.S. Food and Drug Administration (FDA) approved an expanded indication for Verzenio (abemaciclib), in combination with endocrine therapy (ET), for the adjuvant treatment of adult patients with hormone receptor-positive (HR+), human epidermal growth factor receptor 2-negative (HER2-), node-positive, early breast cancer (EBC) at a high risk of recurrence. High risk patients eligible for Verzenio can now be identified solely based on nodal status, tumor size, and tumor grade (4+ positive nodes, or 1-3 positive nodes and at least one of the following: tumors that are ≥5 cm or Grade 3).1 This expanded adjuvant indication removes the Ki-67 score requirement for patient selection.
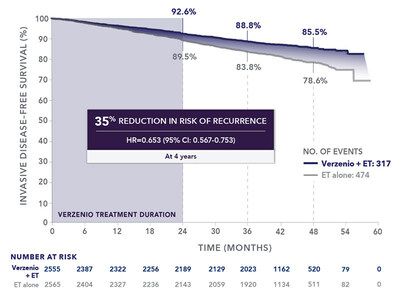
This label expansion is supported by four-year data from the Phase 3 monarchE trial of adjuvant Verzenio in combination with ET, which showed a deepened benefit in invasive disease-free survival (IDFS) beyond the two-year treatment course with adjuvant Verzenio. The absolute difference in IDFS between treatment groups increased over time. At four years, 85.5% of patients remained recurrence-free with Verzenio plus ET, compared to 78.6% with ET alone, an absolute difference in IDFS of 6.9%. At two years and at three years, the absolute differences between treatment groups were 3.1% and 5.0%, respectively; see Fig. 1 below. The addition of Verzenio to ET reduced the risk of recurrence by 35% compared to ET alone (HR=0.653 [95% CI: 0.567-0.753]). There were no new safety findings, and overall results are consistent with the well-established safety profile for Verzenio. These four-year monarchE data were presented at the 2022 San Antonio Breast Cancer Symposium and simultaneously published in The Lancet Oncology.2
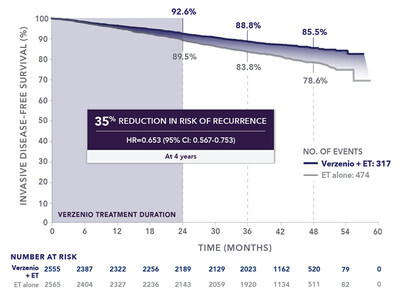
The monarchE study enrolled 5,637 adults with high risk HR+, HER2-, node-positive EBC into two cohorts. Verzenio is now approved for use in the full Cohort 1 patient population, which comprised 91% of the study population. A statistically significant difference in IDFS was observed in the intent-to-treat (ITT) population, primarily due to patients in Cohort 1. As of the data cut-off date, while overall survival (OS) data remain immature across the entire study, an OS trend in favor of Verzenio was observed in the Cohort 1 population, but not the Cohort 2 population where more deaths were seen with Verzenio plus ET compared to ET alone (10/253 vs. 5/264). The “About the monarchE Study” section below provides more details on study design.
“Our goal in intensifying treatment for early breast cancer is to maintain remission and prevent the recurrence of cancer. The magnitude of benefit seen in the four-year data from the monarchE study reinforces my confidence in adjuvant Verzenio as the standard-of-care for high risk patients in this setting,” said Erika P. Hamilton, M.D., medical oncologist, director of Breast and Gynecologic Cancer Research at Sarah Cannon Research Institute, and an investigator on the monarchE clinical trial. “The initial Verzenio FDA approval in early breast cancer was practice-changing and now, through this indication expansion, we have the potential to reduce the risk of breast cancer recurrence for many more patients, relying solely on commonly utilized clinicopathologic features to identify them.”
More than 300,000 people are expected to be diagnosed with breast cancer in the U.S. in 2023.3 It is estimated that 90% of all breast cancers are detected at an early stage.4 Approximately 70% of all breast cancer cases are the HR+, HER2- subtype.5 Although the prognosis for HR+, HER2- EBC is generally favorable, high risk patients are three times more likely than those with low risk characteristics to experience recurrence – with the majority being incurable metastatic disease.6 These patients have an increased risk of recurrence during the first two years of endocrine therapy.
“This expanded approval will allow us to bring Verzenio to many more women and men with HR+, HER2-, high risk early breast cancer in the curative setting – before patients experience recurrence, potentially to incurable metastatic disease,” said Jacob Van Naarden, chief executive officer of Loxo@Lilly. “The initial adjuvant approval for Verzenio changed the treatment paradigm, and the strength of the monarchE results supporting this approval underscores the role this differentiated CDK4/6 inhibitor can play in reducing the risk of recurrence in early breast cancer.”
“This expanded approval for Verzenio is welcome news for our community,” said Jean Sachs, chief executive officer of Living Beyond Breast Cancer. “A significant number of women and men have HR+, HER2- early breast cancer at high risk of returning. Making effective treatment options available is crucial to allowing people to make the best care decisions for themselves, together with their healthcare providers. We’re pleased Verzenio will now be available to more people with this type of early breast cancer.”
Concurrent with this expanded indication approval in EBC, the FDA has also broadened the indicated use of Verzenio in metastatic breast cancer (MBC) when used in combination with an aromatase inhibitor as initial endocrine-based therapy for the treatment of people with HR+, HER2- advanced or MBC. This updated MBC indication now includes all adult patients, with the expanded indication including pre-/perimenopausal women when used in combination with ovarian suppression. See below “Indications for Verzenio” for full details on indicated uses in HR+, HER2- advanced or metastatic breast cancer. Verzenio is available in tablet strengths of 50 mg, 100 mg, 150 mg, and 200 mg.
The labeling for Verzenio contains warnings and precautions for diarrhea, neutropenia, interstitial lung disease (ILD/pneumonitis), hepatotoxicity, venous thromboembolism, and embryo-fetal toxicity. Instruct patients at the first sign of loose stools to initiate antidiarrheal therapy, increase oral fluids, and notify their healthcare provider. Perform complete blood counts and liver function tests prior to the start of Verzenio treatment, every two weeks for the first two months, monthly for the next two months and as clinically indicated. Based on results, Verzenio may require dose modification. Monitor patients for signs and symptoms of thrombosis and pulmonary embolism and treat as medically appropriate. Advise patients of potential risk to a fetus and to use effective contraception.
See Important Safety Information below and full Prescribing Information for additional information.
About the monarchE Study
monarchE was a global, randomized, open-label, two cohort, multicenter Phase 3 clinical trial that enrolled 5,637 adults with HR+, HER2-, node-positive EBC at high risk of recurrence. To be enrolled in Cohort 1 (n=5,120), which is the FDA-approved population, patients had to have 4+ positive nodes or 1-3 positive nodes and at least one of the following: tumors that were ≥5 cm or Grade 3. Patients enrolled in Cohort 2 could not have met the eligibility criteria for Cohort 1. To be enrolled in Cohort 2 (n=517), patients had to have 1-3 positive nodes and Ki-67 score ≥20%. Patients in each cohort were randomized 1:1 to receive either Verzenio 150 mg twice daily plus standard-of-care adjuvant ET (Cohort 1, n=2,555; Cohort 2, n=253) or standard-of-care adjuvant ET alone (Cohort 1, n=2,565; Cohort 2, n=264) for 2 years. ET continued for at least 5 years if deemed medically appropriate. The primary endpoint was IDFS. Consistent with expert guidelines, IDFS was defined as the length of time before breast cancer comes back, any new cancer develops, or death.
About Early Breast Cancer and Risk of Recurrence
It is estimated that 90% of all breast cancers are detected at an early stage.4 Approximately 70% of all breast cancer cases are the HR+, HER2- subtype.5 Although the prognosis for HR+, HER2- EBC is generally favorable, high risk patients are three times more likely than those with low risk characteristics to experience recurrence – with the majority being incurable metastatic disease.6 These patients have an increased risk of recurrence during the first two years of endocrine therapy.
Factors associated with high risk of recurrence in HR+, HER2- early breast cancer include: positive nodal status, the number of positive nodes, large tumor size (≥5 cm), and high tumor grade (Grade 3). Node-positive means that cancer cells from the tumor in the breast have been found in the lymph nodes near the breast. Although breast cancer is removed through surgery, the presence of cancer cells in the lymph nodes signifies that there is a higher chance of developing recurrence and distant metastatic disease.
About Breast Cancer
Breast cancer has surpassed lung cancer as the most commonly diagnosed cancer worldwide, according to GLOBOCAN. The estimated 2.3 million new cases indicate that 1 in every 8 cancers diagnosed in 2020 is breast cancer. With approximately 685,000 deaths in 2020, breast cancer is the fifth-leading cause of cancer death worldwide.7 In the U.S., it is estimated that there will be more than 300,000 new cases of breast cancer diagnosed in 2023. Breast cancer is the second leading cause of cancer death in women in the U.S.3
About Verzenio® (abemaciclib)
Verzenio® (abemaciclib) is a targeted treatment known as a CDK4/6 inhibitor. Verzenio is a nonchemotherapy oral tablet.
Verzenio works inside the cell to block CDK4/6 activity and help stop the growth of cancer cells so that they may eventually die (based on preclinical studies). Cyclin-dependent kinases (CDK)4/6 are activated by binding to D-cyclins. In estrogen receptor-positive (ER+) breast cancer cell lines, cyclin D1 and CDK4/6 promote phosphorylation of the retinoblastoma protein (Rb), cell cycle progression and cell proliferation.
In vitro, continuous exposure to Verzenio inhibited Rb phosphorylation and blocked progression from G1 to S phase of the cell cycle, resulting in senescence and apoptosis (cell death). Preclinically, Verzenio dosed daily without interruption resulted in reduction of tumor size. Inhibiting CDK4/6 in healthy cells can result in side effects, some of which may be serious. Clinical evidence also suggests that Verzenio crosses the blood-brain barrier. In patients with advanced cancer, including breast cancer, concentrations of Verzenio and its active metabolites (M2 and M20) in cerebrospinal fluid are comparable to unbound plasma concentrations.
Verzenio is Lilly’s first solid oral dosage form to be made using a faster, more efficient process known as continuous manufacturing. Continuous manufacturing is a new and advanced type of manufacturing within the pharmaceutical industry, and Lilly is one of the first companies to use this technology.
INDICATIONS FOR VERZENIO®
VERZENIO® is a kinase inhibitor indicated:
IMPORTANT SAFETY INFORMATION FOR VERZENIO (abemaciclib)
Severe diarrhea associated with dehydration and infection occurred in patients treated with Verzenio. Across four clinical trials in 3691 patients, diarrhea occurred in 81 to 90% of patients who received Verzenio. Grade 3 diarrhea occurred in 8 to 20% of patients receiving Verzenio. Most patients experienced diarrhea during the first month of Verzenio treatment. The median time to onset of the first diarrhea event ranged from 6 to 8 days; and the median duration of Grade 2 and Grade 3 diarrhea ranged from 6 to 11 days and 5 to 8 days, respectively. Across trials, 19 to 26% of patients with diarrhea required a Verzenio dose interruption and 13 to 23% required a dose reduction.
Instruct patients to start antidiarrheal therapy, such as loperamide, at the first sign of loose stools, increase oral fluids, and notify their healthcare provider for further instructions and appropriate follow-up. For Grade 3 or 4 diarrhea, or diarrhea that requires hospitalization, discontinue Verzenio until toxicity resolves to ≤Grade 1, and then resume Verzenio at the next lower dose.
Neutropenia, including febrile neutropenia and fatal neutropenic sepsis, occurred in patients treated with Verzenio. Across four clinical trials in 3691 patients, neutropenia occurred in 37 to 46% of patients receiving Verzenio. A Grade ≥3 decrease in neutrophil count (based on laboratory findings) occurred in 19 to 32% of patients receiving Verzenio. Across trials, the median time to first episode of Grade ≥3 neutropenia ranged from 29 to 33 days, and the median duration of Grade ≥3 neutropenia ranged from 11 to 16 days. Febrile neutropenia has been reported in <1% of patients exposed to Verzenio across trials. Two deaths due to neutropenic sepsis were observed in MONARCH 2. Inform patients to promptly report any episodes of fever to their healthcare provider.
Monitor complete blood counts prior to the start of Verzenio therapy, every 2 weeks for the first 2 months, monthly for the next 2 months, and as clinically indicated. Dose interruption, dose reduction, or delay in starting treatment cycles is recommended for patients who develop Grade 3 or 4 neutropenia.
Severe, life-threatening, or fatal interstitial lung disease (ILD) or pneumonitis can occur in patients treated with Verzenio and other CDK4/6 inhibitors. In Verzenio-treated patients in EBC (monarchE), 3% of patients experienced ILD or pneumonitis of any grade: 0.4% were Grade 3 or 4 and there was one fatality (0.1%). In Verzenio-treated patients in MBC (MONARCH 1, MONARCH 2, MONARCH 3), 3.3% of Verzenio-treated patients had ILD or pneumonitis of any grade: 0.6% had Grade 3 or 4, and 0.4% had fatal outcomes. Additional cases of ILD or pneumonitis have been observed in the postmarketing setting, with fatalities reported.
Monitor patients for pulmonary symptoms indicative of ILD or pneumonitis. Symptoms may include hypoxia, cough, dyspnea, or interstitial infiltrates on radiologic exams. Infectious, neoplastic, and other causes for such symptoms should be excluded by means of appropriate investigations. Dose interruption or dose reduction is recommended in patients who develop persistent or recurrent Grade 2 ILD or pneumonitis. Permanently discontinue Verzenio in all patients with Grade 3 or 4 ILD or pneumonitis.
Grade ≥3 increases in alanine aminotransferase (ALT) (2 to 6%) and aspartate aminotransferase (AST) (2 to 3%) were reported in patients receiving Verzenio. Across three clinical trials in 3559 patients (monarchE, MONARCH 2, MONARCH 3), the median time to onset of Grade ≥3 ALT increases ranged from 57 to 87 days and the median time to resolution to Grade <3 was 13 to 14 days. The median time to onset of Grade ≥3 AST increases ranged from 71 to 185 days and the median time to resolution to Grade <3 ranged from 11 to 15 days.
Monitor liver function tests (LFTs) prior to the start of Verzenio therapy, every 2 weeks for the first 2 months, monthly for the next 2 months, and as clinically indicated. Dose interruption, dose reduction, dose discontinuation, or delay in starting treatment cycles is recommended for patients who develop persistent or recurrent Grade 2, or any Grade 3 or 4 hepatic transaminase elevation.
Venous thromboembolic events (VTE) were reported in 2 to 5% of patients across three clinical trials in 3559 patients treated with Verzenio (monarchE, MONARCH 2, MONARCH 3). VTE included deep vein thrombosis, pulmonary embolism, pelvic venous thrombosis, cerebral venous sinus thrombosis, subclavian and axillary vein thrombosis, and inferior vena cava thrombosis. In clinical trials, deaths due to VTE have been reported in patients treated with Verzenio.
Verzenio has not been studied in patients with early breast cancer who had a history of VTE. Monitor patients for signs and symptoms of venous thrombosis and pulmonary embolism and treat as medically appropriate. Dose interruption is recommended for EBC patients with any grade VTE and for MBC patients with a Grade 3 or 4 VTE.
Verzenio can cause fetal harm when administered to a pregnant woman, based on findings from animal studies and the mechanism of action. In animal reproduction studies, administration of abemaciclib to pregnant rats during the period of organogenesis caused teratogenicity and decreased fetal weight at maternal exposures that were similar to the human clinical exposure based on area under the curve (AUC) at the maximum recommended human dose. Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective contraception during treatment with Verzenio and for 3 weeks after the last dose. Based on findings in animals, Verzenio may impair fertility in males of reproductive potential. There are no data on the presence of Verzenio in human milk or its effects on the breastfed child or on milk production. Advise lactating women not to breastfeed during Verzenio treatment and for at least 3 weeks after the last dose because of the potential for serious adverse reactions in breastfed infants.
The most common adverse reactions (all grades, ≥10%) observed in monarchE for Verzenio plus tamoxifen or an aromatase inhibitor vs tamoxifen or an aromatase inhibitor, with a difference between arms of ≥2%, were diarrhea (84% vs 9%), infections (51% vs 39%), neutropenia (46% vs 6%), fatigue (41% vs 18%), leukopenia (38% vs 7%), nausea (30% vs 9%), anemia (24% vs 4%), headache (20% vs 15%), vomiting (18% vs 4.6%), stomatitis (14% vs 5%), lymphopenia (14% vs 3%), thrombocytopenia (13% vs 2%), decreased appetite (12% vs 2.4%), ALT increased (12% vs 6%), AST increased (12% vs 5%), dizziness (11% vs 7%), rash (11% vs 4.5%), and alopecia (11% vs 2.7 %).
The most frequently reported ≥5% Grade 3 or 4 adverse reaction that occurred in the Verzenio arm vs the tamoxifen or an aromatase inhibitor arm of monarchE were neutropenia (19.6% vs 1%), leukopenia (11% vs <1%), diarrhea (8% vs 0.2%), and lymphopenia (5% vs <1%).
Lab abnormalities (all grades; Grade 3 or 4) for monarchE in ≥10% for Verzenio plus tamoxifen or an aromatase inhibitor with a difference between arms of ≥2% were increased serum creatinine (99% vs 91%; .5% vs <.1%), decreased white blood cells (89% vs 28%; 19.1% vs 1.1%), decreased neutrophil count (84% vs 23%; 18.7% vs 1.9%), anemia (68% vs 17%; 1% vs .1%), decreased lymphocyte count (59% vs 24%; 13.2 % vs 2.5%), decreased platelet count (37% vs 10%; .9% vs .2%), increased ALT (37% vs 24%; 2.6% vs 1.2%), increased AST (31% vs 18%; 1.6% vs .9%), and hypokalemia (11% vs 3.8%; 1.3% vs 0.2%).
The most common adverse reactions (all grades, ≥10%) observed in MONARCH 3 for Verzenio plus anastrozole or letrozole vs anastrozole or letrozole, with a difference between arms of ≥2%, were diarrhea (81% vs 30%), fatigue (40% vs 32%), neutropenia (41% vs 2%), infections (39% vs 29%), nausea (39% vs 20%), abdominal pain (29% vs 12%), vomiting (28% vs 12%), anemia (28% vs 5%), alopecia (27% vs 11%), decreased appetite (24% vs 9%), leukopenia (21% vs 2%), creatinine increased (19% vs 4%), constipation (16% vs 12%), ALT increased (16% vs 7%), AST increased (15% vs 7%), rash (14% vs 5%), pruritus (13% vs 9%), cough (13% vs 9%), dyspnea (12% vs 6%), dizziness (11% vs 9%), weight decreased (10% vs 3.1%), influenza-like illness (10% vs 8%), and thrombocytopenia (10% vs 2%).
The most frequently reported ≥5% Grade 3 or 4 adverse reactions that occurred in the Verzenio arm vs the placebo arm of MONARCH 3 were neutropenia (22% vs 1%), diarrhea (9% vs 1.2%), leukopenia (7% vs <1%)), increased ALT (6% vs 2%), and anemia (6% vs 1%).
Lab abnormalities (all grades; Grade 3 or 4) for MONARCH 3 in ≥10% for Verzenio plus anastrozole or letrozole with a difference between arms of ≥2% were increased serum creatinine (98% vs 84%; 2.2% vs 0%), decreased white blood cells (82% vs 27%; 13% vs 0.6%), anemia (82% vs 28%; 1.6% vs 0%), decreased neutrophil count (80% vs 21%; 21.9% vs 2.6%), decreased lymphocyte count (53% vs 26%; 7.6% vs 1.9%), decreased platelet count (36% vs 12%; 1.9% vs 0.6%), increased ALT (48% vs 25%; 6.6% vs 1.9%), and increased AST (37% vs 23%; 3.8% vs 0.6%).
The most common adverse reactions (all grades, ≥10%) observed in MONARCH 2 for Verzenio plus fulvestrant vs fulvestrant, with a difference between arms of ≥2%, were diarrhea (86% vs 25%), neutropenia (46% vs 4%), fatigue (46% vs 32%), nausea (45% vs 23%), infections (43% vs 25%), abdominal pain (35% vs 16%), anemia (29% vs 4%), leukopenia (28% vs 2%), decreased appetite (27% vs 12%), vomiting (26% vs 10%), headache (20% vs 15%), dysgeusia (18% vs 2.7%), thrombocytopenia (16% vs 3%), alopecia (16% vs 1.8%), stomatitis (15% vs 10%), ALT increased (13% vs 5%), pruritus (13% vs 6%), cough (13% vs 11%), dizziness (12% vs 6%), AST increased (12% vs 7%), peripheral edema (12% vs 7%), creatinine increased (12% vs <1%), rash (11% vs 4.5%), pyrexia (11% vs 6%), and weight decreased (10% vs 2.2%).
The most frequently reported ≥5% Grade 3 or 4 adverse reactions that occurred in the Verzenio arm vs the placebo arm of MONARCH 2 were neutropenia (25% vs 1%), diarrhea (13% vs 0.4%), leukopenia (9% vs 0%), anemia (7% vs 1%), and infections (5.7% vs 3.5%).
Lab abnormalities (all grades; Grade 3 or 4) for MONARCH 2 in ≥10% for Verzenio plus fulvestrant with a difference between arms of ≥2% were increased serum creatinine (98% vs 74%; 1.2% vs 0%), decreased white blood cells (90% vs 33%; 23.7% vs .9%), decreased neutrophil count (87% vs 30%; 32.5% vs 4.2%), anemia (84% vs 34%; 2.6% vs .5%), decreased lymphocyte count (63% vs 32%; 12.2% vs 1.8%), decreased platelet count (53% vs 15%; 2.1% vs 0%), increased ALT (41% vs 32%; 4.6% vs 1.4%), and increased AST (37% vs 25%; 3.9% vs 4.2%).
The most common adverse reactions (all grades, ≥10%) observed in MONARCH 1 with Verzenio were diarrhea (90%), fatigue (65%), nausea (64%), decreased appetite (45%), abdominal pain (39%), neutropenia (37%), vomiting (35%), infections (31%), anemia (25%), thrombocytopenia (20%), headache (20%), cough (19%), constipation (17%), leukopenia (17%), arthralgia (15%), dry mouth (14%), weight decreased (14%), stomatitis (14%), creatinine increased (13%), alopecia (12%), dysgeusia (12%), pyrexia (11%), dizziness (11%), and dehydration (10%).
The most frequently reported ≥5% Grade 3 or 4 adverse reactions from MONARCH 1 with Verzenio were diarrhea (20%), neutropenia (24%), fatigue (13%), and leukopenia (5%).
Lab abnormalities (all grades; Grade 3 or 4) for MONARCH 1 with Verzenio were increased serum creatinine (99%; .8%), decreased white blood cells (91%; 28%), decreased neutrophil count (88%; 26.6%), anemia (69%; 0%), decreased lymphocyte count (42%; 13.8%), decreased platelet count (41%; 2.3%), increased ALT (31%; 3.1%), and increased AST (30%; 3.8%).
Strong and moderate CYP3A inhibitors increased the exposure of abemaciclib plus its active metabolites to a clinically meaningful extent and may lead to increased toxicity. Avoid concomitant use of ketoconazole. Ketoconazole is predicted to increase the AUC of abemaciclib by up to 16-fold. In patients with recommended starting doses of 200 mg twice daily or 150 mg twice daily, reduce the Verzenio dose to 100 mg twice daily with concomitant use of strong CYP3A inhibitors other than ketoconazole. In patients who have had a dose reduction to 100 mg twice daily due to adverse reactions, further reduce the Verzenio dose to 50 mg twice daily with concomitant use of strong CYP3A inhibitors. If a patient taking Verzenio discontinues a strong CYP3A inhibitor, increase the Verzenio dose (after 3 to 5 half-lives of the inhibitor) to the dose that was used before starting the inhibitor. With concomitant use of moderate CYP3A inhibitors, monitor for adverse reactions and consider reducing the Verzenio dose in 50 mg decrements. Patients should avoid grapefruit products.
Avoid concomitant use of strong or moderate CYP3A inducers and consider alternative agents. Coadministration of strong or moderate CYP3A inducers decreased the plasma concentrations of abemaciclib plus its active metabolites and may lead to reduced activity.
With severe hepatic impairment (Child-Pugh C), reduce the Verzenio dosing frequency to once daily. The pharmacokinetics of Verzenio in patients with severe renal impairment (CLcr <30 mL/min), end stage renal disease, or in patients on dialysis is unknown. No dosage adjustments are necessary in patients with mild or moderate hepatic (Child-Pugh A or B) and/or renal impairment (CLcr ≥30-89 mL/min).
Please see full Prescribing Information for Verzenio.
AL HCP ISI 12OCT2021
About Lilly
Lilly unites caring with discovery to create medicines that make life better for people around the world. We’ve been pioneering life-changing discoveries for nearly 150 years, and today our medicines help more than 47 million people across the globe. Harnessing the power of biotechnology, chemistry and genetic medicine, our scientists are urgently advancing new discoveries to solve some of the world’s most significant health challenges, redefining diabetes care, treating obesity and curtailing its most devastating long-term effects, advancing the fight against Alzheimer’s disease, providing solutions to some of the most debilitating immune system disorders, and transforming the most difficult-to-treat cancers into manageable diseases. With each step toward a healthier world, we’re motivated by one thing: making life better for millions more people. That includes delivering innovative clinical trials that reflect the diversity of our world and working to ensure our medicines are accessible and affordable. To learn more, visit Lilly.com and Lilly.com/newsroom or follow us on Facebook, Instagram and LinkedIn.
Cautionary Statement Regarding Forward-Looking Statements
This press release contains forward-looking statements (as that term is defined in the Private Securities Litigation Reform Act of 1995) about Verzenio (abemaciclib) as a treatment for people with early breast cancer and milestones relating to Verzenio, and reflects Lilly’s current beliefs and expectations. However, as with any pharmaceutical product, there are substantial risks and uncertainties in the process of drug research, development, and commercialization. Among other things, there is no guarantee that planned or ongoing studies will be completed as planned, that future study results will be consistent with study results to date, that Verzenio will receive additional regulatory approvals, or that Lilly will execute its strategy as expected. For further discussion of these and other risks and uncertainties that could cause actual results to differ from Lilly’s expectations, see Lilly’s Form 10-K and Form 10-Q filings with the United States Securities and Exchange Commission. Except as required by law, Lilly undertakes no duty to update forward-looking statements to reflect events after the date of this release.
1 Verzenio. Prescribing information. Lilly USA, LLC.
2 Johnston SRD, Toi M, O’Shaughnessy J, Rastogi P, et al. Abemaciclib plus endocrine therapy for hormone receptor-positive, HER2-negative, node-positive, high-risk early breast cancer (monarchE): results from a preplanned interim analysis of a randomised, open-label, phase 3 trial. Lancet Oncol. 2023 Jan;24(1):77-90.
3 American Cancer Society. Cancer Statistics Center. http://cancerstatisticscenter.cancer.org. Accessed February 24, 2023.
4 Howlander N, et al. SEER Cancer Statistics Review, 1975-2017, based on November 2019 SEER data submission. https://seer.cancer.gov/archive/csr/1975_2017/. Accessed February 24, 2023.
5 National Cancer Institute, SEER. Cancer Stat Facts: Female Breast Cancer. https://seer.cancer.gov/statfacts/html/breast.html. Accessed February 24, 2023.
6 Early Breast Cancer Trialists’ Collaborative Group (EBCTCG). Effects of chemotherapy and hormonal therapy for early breast cancer on recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;365(9472):1687-1717. doi:10.1016/S0140-6736(05)66544-0.
7 Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71(3):209-249.
SOURCE Eli Lilly and Company
Posted: March 2023
Related articles
- FDA Approves Verzenio (abemaciclib) as the First and Only CDK4/6 Inhibitor for Certain People with HR+ HER2- High Risk Early Breast Cancer – October 13, 2021
- Lilly Receives Additional FDA Approval for Verzenio (abemaciclib), as Initial Treatment for Advanced Breast Cancer – February 26, 2018
- FDA Approves Verzenio (abemaciclib) for Certain Advanced or Metastatic Breast Cancers – September 28, 2017
- FDA Grants Priority Review for Lilly’s Abemaciclib for the Treatment of Advanced Breast Cancer – July 12, 2017
Verzenio (abemaciclib) FDA Approval History
Source: Read Full Article